Month: March 2015
-
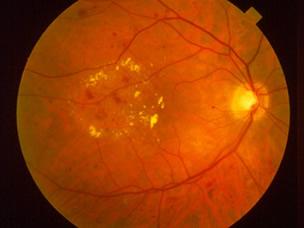
FDA Approves New Treatment for Diabetic Retinopathy in Patients
Diabetic retinopathy (DR) is the most common diabetic eye disease and is a leading cause of blindness in adults in the United States…
-
Traffic-Related Air Pollution Linked to Type 1 Diabetes in Children
A Munich study now shows that high levels of fine dust pollution could possibly increase the risk of type 1 diabetes among children…
-
Novel Drug Candidate Regenerates Pancreatic Cells Lost in Diabetes
The Mount Sinai study found that harmine drove the sustained division and multiplication of adult human beta cells in culture…
-
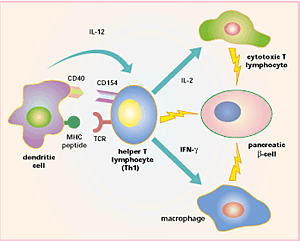
Genes That Increase the Risk of Type 1 Diabetes Have Lost Their Hiding Place
Newswise — The genes that increase the risk of type 1 diabetes have lost their hiding place…
-
Type 1 Diabetes Originates in the Gut But Probiotics Could Offer Cure
[ New research that ] development of type 1 diabetes is likely caused by the gut, and therefore, a type of probiotic could be the cure…
-
Children in UK: Alarming Rise in Type 1 Diabetes Among Youth
[ Many children ] in the UK have been found to exhibit long-term health implications associated with type 1 diabetes…
-
Young Scientists Face The Worst Research Funding In 50 Years
Young scientists entering biomedical research find themselves in the worst financial environment in half a century…
-
New Crowdfunding Hub to Choose and Follow Your Donations
Each time I donate to diabetes research in recent years, I get this sinking feeling that I’m throwing money into a bottomless pit…